The Business Research Company’s Viral Vector CDMO Market to Hit $2.58 Billion by 2030, Growing at a 15.3% CAGR
LONDON, GREATER LONDON, UNITED KINGDOM, May 1, 2026 /EINPresswire.com/ — “Viral Vector Contract Development And Manufacturing Organization (CDMO) market to surpass $3 billion in 2030. In comparison, the Pharmaceutical Contract Development And Manufacturing (CDMO) market, which is considered as its parent market, is expected to be approximately $281 billion by 2030, with Viral Vector Contract Development And Manufacturing Organization (CDMO) to represent around 1% of the parent market. Within the broader Pharmaceuticals industry, which is expected to be $2,513 billion by 2030, the Viral Vector Contract Development And Manufacturing Organization (CDMO) market is estimated to account for nearly 0.1% of the total market value.
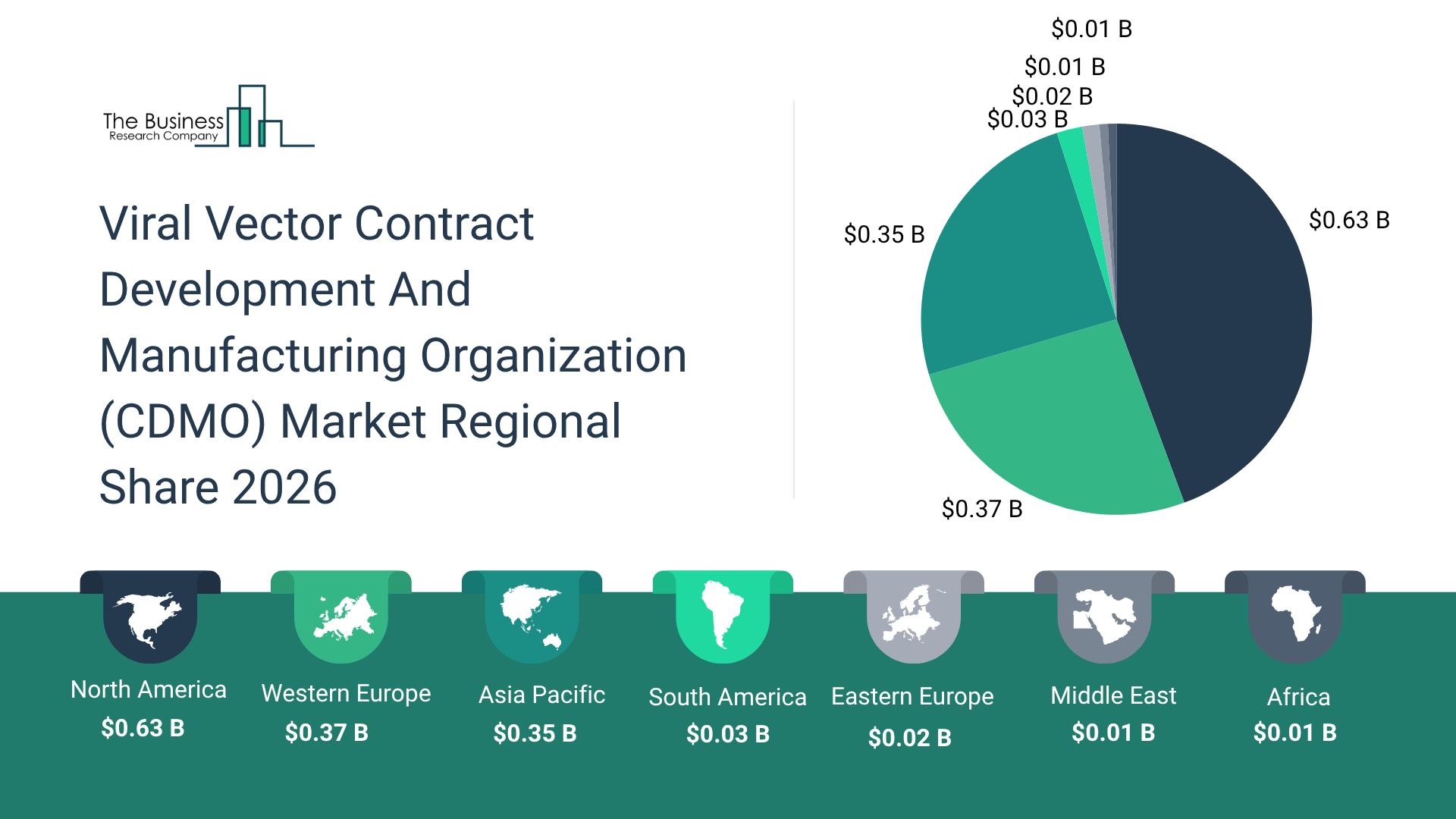
Which Will Be The Biggest Region In The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market In 2030?
North America will be the largest region in the viral vector contract development and manufacturing organization (CDMO) market in 2030, valued at $1.1 billion. The market is expected to grow from $0.6 billion in 2025 at a compound annual growth rate (CAGR) of 14%. The rapid growth can be attributed to the presence of leading gene therapy developers and CDMOs across the USA, increasing clinical pipeline of gene and cell therapies, rising demand for high-quality GMP viral vector manufacturing, strong investments in advanced biomanufacturing infrastructure, and growing partnerships between biotech firms and CDMOs to accelerate therapy commercialization within the region.
Which Will Be The Largest Country In The Global Viral Vector Contract Development And Manufacturing Organization (CDMO) Market In 2030?
The USA will be the largest country in the viral vector contract development and manufacturing organization (CDMO) market in 2030, valued at $1.0 billion. The market is expected to grow from $0.5 billion in 2025 at a compound annual growth rate (CAGR) of 14%. The rapid growth can be attributed to a robust biotechnology ecosystem, high concentration of clinical trials for gene and cell therapies, increasing outsourcing of viral vector manufacturing, rapid expansion of GMP-certified production facilities, and continuous innovation in vector engineering and scalable manufacturing technologies by domestic players.
Request A Free Sample Of The Viral Vector Contract Development and Manufacturing Organization (CDMO) Market Report
https://www.thebusinessresearchcompany.com/sample_request?id=29238&type=smp&utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Apr_PR
What Will Be The Largest Segment In The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market In 2030?
The viral vector contract development and manufacturing organization (CDMO) market is segmented by service type into process development, manufacturing, analytical testing, fill-finish, and other service types. The manufacturing segment will be the largest segment of the viral vector contract development and manufacturing organization (CDMO) market segmented by service type, accounting for 44% or $1.1 billion of the total in 2030. The manufacturing market will be supported by the growing demand for large-scale GMP production of viral vectors, increasing number of late-stage clinical and commercial gene therapy programs, high capital requirements for in-house manufacturing, rising reliance on CDMOs for scalable production, and continuous advancements in bioprocessing technologies to improve yield, quality, and cost-efficiency.
The viral vector contract development and manufacturing organization (CDMO) market is segmented by vector type into adenoviral vectors, lentiviral vectors, adeno-associated viral vectors, retroviral vectors, and other vector types.
The viral vector contract development and manufacturing organization (CDMO) market is segmented by workflow into upstream manufacturing and downstream manufacturing.
The viral vector contract development and manufacturing organization (CDMO) market is segmented by application into gene therapy, vaccines, cell therapy, and other applications.
The viral vector contract development and manufacturing organization (CDMO) market is segmented by end-user into pharmaceutical and biotechnology companies, academic and research institutes, and other end-users.
What Is The Expected CAGR For The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Leading Up To 2030?
The expected CAGR for the viral vector contract development and manufacturing organization (CDMO) market leading up to 2030 is 15%.
What Will Be The Growth Driving Factors In The Global Viral Vector Contract Development And Manufacturing Organization (CDMO) Market In The Forecast Period?
The rapid growth of the global viral vector contract development and manufacturing organization (CDMO) market leading up to 2030 will be driven by the following key factors including expanding outsourced manufacturing capacity and CDMO investments, stricter and harmonized regulatory standards, rising demand for personalized and autologous manufacturing, advancements in scalable vector production technologies, and increasing strategic partnerships between biotech firms and CDMOs.
Rapid Expansion Of Outsourced Capacity And CDMO Investments – The rapid expansion of outsourced capacity and CDMO investments is expected to become a key growth driver for the viral vector CDMO market by 2030. Biopharmaceutical companies are increasingly outsourcing viral vector development and manufacturing to specialized CDMOs to reduce capital expenditure, accelerate timelines, and access advanced bioprocessing expertise. Significant investments in facility expansions, single-use technologies, and large-scale GMP manufacturing suites are enhancing global production capacity. This trend is enabling faster progression from clinical development to commercialization while ensuring supply reliability. As a result, the rapid expansion of outsourced capacity and CDMO investments is anticipated to contribute approximately 3.0% annual growth to the market.
Tighter Regulatory Expectations And Quality/Regulatory Harmonization – Tighter regulatory expectations and quality and regulatory harmonization are expected to emerge as major factors driving the expansion of the viral vector CDMO market by 2030. Increasing scrutiny from global regulatory agencies is pushing CDMOs to adopt standardized GMP practices, robust quality systems, and validated manufacturing processes. Harmonization of regulatory guidelines across regions is further encouraging the development of globally compliant manufacturing platforms. Companies are investing in quality assurance infrastructure and regulatory expertise to ensure consistent product quality and faster approvals. Consequently, tighter regulatory expectations and harmonization are projected to contribute around 2.8% annual growth to the market.
Demand For On-Demand/Personalized And Autologous Manufacturing – The growing demand for on-demand, personalized, and autologous manufacturing is expected to act as a key growth catalyst for the viral vector CDMO market by 2030. The rise of patient-specific therapies, particularly in cell and gene therapy, requires flexible, small-batch, and highly controlled manufacturing processes. CDMOs are developing modular and decentralized manufacturing platforms to support rapid turnaround times and individualized treatment production. This shift is increasing the need for adaptable manufacturing infrastructure and advanced process control systems. Therefore, the demand for on-demand and autologous manufacturing is projected to contribute approximately 2.5% annual growth to the market.
Access The Detailed Viral Vector Contract Development And Manufacturing Organization (CDMO) Market Report Here
https://www.thebusinessresearchcompany.com/report/viral-vector-contract-development-and-manufacturing-organization-cdmo-global-market-report?utm_source=EINPresswire&utm_medium=Paid&utm_campaign=Apr_PR
What Are The Key Growth Opportunities In The Viral Vector Contract Development And Manufacturing Organization (CDMO) Market In 2030?
The most significant growth opportunities are anticipated in the process development, manufacturing, analytical testing, fill-finish, and other service types market. Collectively, these segments are projected to contribute over $1.3 billion in market value by 2030, driven by the increasing number of gene and cell therapy pipelines, rising demand for scalable GMP viral vector production, growing reliance on CDMOs for specialized development and manufacturing capabilities, advancements in bioprocessing and analytical technologies, and the need for integrated end-to-end service offerings. This momentum reflects the biopharmaceutical industry’s focus on accelerating clinical timelines, ensuring regulatory compliance, and enabling efficient commercialization of advanced therapies, thereby driving growth across the global viral vector CDMO ecosystem.
The process development market is projected to grow by $0.4 billion, the manufacturing market by $0.6 billion, the analytical testing market by $0.2 billion, the fill-finish market by $0.1 billion, and the other service types market by $0.1 billion over the next five years from 2025 to 2030.
Learn More About The Business Research Company
The Business Research Company (www.thebusinessresearchcompany.com) is a leading market intelligence firm renowned for its expertise in company, market, and consumer research. We have published over 17,500 reports across 27 industries and 60+ geographies. Our research is powered by 1,500,000 datasets, extensive secondary research, and exclusive insights from interviews with industry leaders.
We provide continuous and custom research services, offering a range of specialized packages tailored to your needs, including Market Entry Research Package, Competitor Tracking Package, Supplier & Distributor Package and much more.
Disclaimer: Please note that the findings, conclusions and recommendations that TBRC Business Research Pvt Ltd delivers are based on information gathered in good faith from both primary and secondary sources, whose accuracy we are not always in a position to guarantee. As such TBRC Business Research Pvt Ltd can accept no liability whatever for actions taken based on any information that may subsequently prove to be incorrect. Analysis and findings included in TBRC reports and presentations are our estimates, opinions and are not intended as statements of fact or investment guidance.”
Oliver Guirdham
The Business Research Company
+44 7882 955267
info@tbrc.info
Visit us on social media:
LinkedIn
Facebook
X
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()
Media gallery